Vaccinating theImmunocompromised Child
Given the array of childhood disorders that can adversely affect the immune system, most pediatricians at some point will care for children who are immunodeficient.
Given the array of childhood disorders that can adversely affect the immune system, most pediatricians at some point will care for children who are immunodeficient. The well-child care of these patients, just like that for immunocompetent children, includes counseling parents about routine vaccines.1-9
In this article, we discuss the basic principles (as articulated by the American Academy of Pediatrics [AAP]5,6 and other sources1,2,5,6,10-17) that guide decisions about how best to protect immunocompromised children against vaccine-preventable illnesses. Although we could not address every possible immunodeficiency state, we have included a variety of important and representative examples. Keep in mind that the unique medical circumstances of each immunocompromised child need to be considered to determine the optimal individual vaccination plan.
PREVALENCE OF CHILDHOOD IMMUNODEFICIENCY ON THE RISE
Primary immunodeficiency diseases (inherited defects of humoral or cellular immunity and defects of phagocytes and the complement system) occur in an estimated 1 in 10,000 persons.18 On the basis of telephone interviews, Boyle and Buckley19 report a calculated prevalence of primary immunodeficiency diseases (such as common variable immunodeficiency, IgA and IgG subclass deficiencies, and others) of 1 in 2000 in US children.
Data indicate that the number of children becoming immunocompromised as a result of secondary conditions (infections, cancer, use of medications, malnutrition, diabetes, kidney disease, and so forth) is increasing. Between the start of the AIDS epidemic and 2007, AIDS was diagnosed in approximately 10,000 children younger than 13 years in the United States.20 The number of US children who received an organ transplant in a given year increased 13% from 1995 to 2004, such that in 2004, there were almost 2000 pediatric transplant recipients.21 The American Cancer Society predicts that in 2009, cancer will be diagnosed in about 10,730 US children younger than 15 years.22 In addition, for all childhood cancers combined, the 5- year relative survival has increased from less than 50% before the 1970s to nearly 80% today13; this markedly improved survival has further expanded the population of immunocompromised children. Some children in whom cancer develops may have already been vaccinated; however, because of medical procedures such as allogenic bone marrow transplantation, preexisting immunities may be attenuated or lost.
Eventually, vaccination regimens need to be established for all of these immunocompromised children.
VACCINATIONS AND IMMUNOCOMPROMISED CHILDREN: THE BASICSDetermine Immune Status1 To clarify a given child's immune status, his or her underlying diagnoses need to be considered and the degree of immunosuppression must be determined. You may want to consult an immunologist to aid in the immunological evaluation and in development of the vaccination plan. Other medical specialists, such as a hematologist-oncologist, may also provide helpful suggestions.
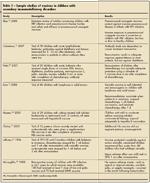
Perform Careful Assessment of Risks Versus Benefits2 The risks and benefits of vaccinating an immunocompromised child have to be considered before any vaccines are administered.1 There have been many studies of vaccine safety and efficacy in immunocompromised children10-17; several representative studies involving secondary immunodeficiency conditions are listed in Table 1. However, because there is such diversity in the primary and secondary conditions that can lead to immunosuppression, each child will need to be considered individually in order to determine his actual risk of acquiring natural infection and of experiencing complications from such an infection-as well as the risk associated with vaccine administration.
Only then can the risks of vaccination be accurately weighed against those of not vaccinating.
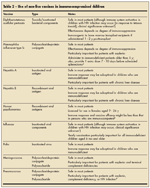
Consider Inactivated Vaccines 3 Inactivated, or non-live, vaccines (Table 2) are generally safe in children with primary and secondary immunodeficiencies, although a modified schedule may be necessary. However, while they are safe, inactivated vaccines may not incite an effective immune response in these children.
All vaccines are probably ineffective in children with severe immunosuppression, such as those with severe combined immunodeficiency or complete DiGeorge syndrome.6 In children who are unable to mount an antibody immune response, passive immunization with intravenous immunoglobulin (IVIG) is the main therapeutic intervention for preventing infections.
Attenuated immune responses following vaccination are likely in children with less severe disorders, such as selective IgA deficiency or IgG subclass deficiencies. However, because the immune response may not be adequate and immunity cannot be assumed, measurement of prevaccination and postvaccination antibody titers may be helpful.23 Consultation with an immunologist is helpful in such situations.
In patients with complement deficiencies or disorders of phagocyte numbers or function, all routine vaccines are probably effective.6 Although all inactivated vaccines are important, some are especially so in children with certain primary deficiencies (for example, the meningococcal vaccine in patients with terminal complement deficiencies and the pneumococcal and meningococcal vaccines in patients with asplenia).
The effectiveness of inactivated vaccines in children with secondary immunodeficiency states varies; it is dependent on both the degree of immunosuppression and the underlying condition with which the immunosuppression is associated. Immunity to infection is not always guaranteed postvaccination. For example, administration of tetanus immunoglobulin is necessary if a child with HIV infection experiences a tetanus-prone wound, whether or not the child has been vaccinated.5
Special rules govern vaccine administration in patients who are immunosuppressed as a result of medical interventions. In children who are receiving immunosuppressive agents or chemotherapy, the routine immunization schedule may need to be adjusted. In a child receiving immunosuppressive agents, vaccination should occur at least 3 months after the agents have been stopped; the amount of time required for an adequate immune response to occur varies depending on the specifics of the earlier treatment regimen. Various vaccination and revaccination strategies have been developed for patients who have had chemotherapy or a bone marrow transplant.11,12
In patients with HIV infection, some vaccines (influenza, tetanus, and pneumococcal polysaccharide) have been associated with an increase in viremia. However, the exact clinical significance of this phenomenon is unknown.1

For Certain Children, Consider Live Vaccines for Which Safety and Efficacy Data Are Available 4 The live vaccines routinely recommended in the United States are the measlesmumps- rubella (MMR) vaccine, the varicella vaccine, the live attenuated influenza vaccine (LAIV), and the rotavirus vaccine (Table 3). The manufacturers of the rotavirus vaccine licensed for use in the United States warn that no safety or efficacy data are available for its administration to infants who are potentially immunocompromised, including those receiving immunosuppressive therapy.8 However, safety and efficacy data are available for the MMR and varicella vaccines. Thus, these vaccines can be administered to some children with primary or secondary immunodeficiency states, depending on the patient's immune status.
All live vaccines are contraindicated in patients with severe immunodeficiency disorders or T-cell immunodeficiency disorders, such as severe combined immunodeficiency syndrome, DiGeorge syndrome, Wiskott-Aldrich syndrome, and ataxia telangectasia.6 It is important to emphasize that postvaccination immunity is not guaranteed in immunocompromised children. Thus, it is recommended that immunoglobulin be administered to children with HIV infection and those who are severely immunocompromised, regardless of immunization status, if they have been exposed to measles or varicella. 5 If a child has received immunoglobulin as a treatment for another disease, live vaccines must be delayed for 6 months.24
Antibody deficiencies. In children who have conditions associated with antibody deficiencies, such as X-linked agammaglobulinemia, measles vaccination can be considered.6 However, these children may not develop antibody responses to vaccines. Also, these patients are protected by the specific antibodies they receive through the IVIG they are given; thus, vaccination may be unnecessary.
HIV infection. In children with HIV infection aged 1 to 8 years who are in CDC clinical category N, A, or B, the MMR vaccine is recommended. Varicella vaccine may be considered in children with HIV infection if their CD4+ T-lymphocyte percentage is 15% or greater. For children with HIV infection who are older than 9 years, consider the MMR and varicella vaccines if the CD4+ cell counts are greater than 200/µL. Although the MMR and varicella vaccines should not be given to infants with HIV infection, the rotavirus vaccine may be administered both to infants infected with HIV and to infants who have been exposed to HIV.
Complement and neutrophil dysfunction. In patients with complement and neutrophil dysfunction, live virus vaccines are considered safe. However, live bacteria vaccines, such as BCG and the live typhoid vaccine that contains Ty21a Salmonella typhi, are contraindicated in patients with neutrophil dysfunction.
Iatrogenic immunosuppression. In patients with iatrogenic immunosuppression, immunization with live vaccines is most successful when vaccination is completed before initiation of the immune-modifying therapy or when the immunosuppressive agents have been temporarily discontinued.25 When it comes to vaccinating the child who will need to begin aggressive therapy with immune-modifying medication, forethought is crucial. Levin25 states that the ideal window of opportunity for vaccinating children who will be receiving immunosuppressive therapy varies depending on the immune-modifying treatment regimen that they will receive.
Systemic corticosteroid treatment. Children who have been receiving systemic corticosteroids for more than 14 days (at a dosage of more than 2 mg/kg/d or with a total daily dose of more than 20 mg of prednisone in a child who weighs more than 10 kg) are considered immunosuppressed. 6 In a child who has received corticosteroids for 2 weeks or longer, live vaccines can be administered no earlier than 3 months after discontinuation of the corticosteroids. The Red Book lists other corticosteroid treatment settings.6
Vaccinate Contacts5 All close contacts who are eligible for vaccinations should be maximally vaccinated; they should receive all routine recommended vaccines, including the live attenuated MMR, varicella, and rotavirus vaccines. 6 Good hand washing is always essential, but particularly so when the rotavirus vaccine has been administered. Viral shedding in recipients of the rotavirus vaccine occurs as early as 1 day after vaccination and as late as 15 days after a dose8; thus, good hand washing is essential for anyone who cares for infants who have received the rotavirus vaccine. With respect to LAIV, limited data are available regarding transmission of the virus from vaccinated persons to immunocompromised patients; however, transmission is thought to occur rarely. It is very important to administer the influenza vaccine annually to all contacts of immunocompromised patients.
Know the Current Vaccine Recommendations6 The pediatric health care provider is referred to the AAP and the CDC for the most up-to-date recommendations on vaccinations in all patients. Recommendations concerning the immunization of immunocompromised children change frequently as more studies about safety and efficacy and new vaccines become available.
References:
REFERENCES:
1. Pirofski LA, Casadevall A. Use of licensedvaccines for active immunization of the immunocompromisedhost.
Clin Microbiol Rev
. 1998;11:1-26.
2. American Academy of Pediatrics.
Haemophilusinfluenzae
infections. In: Pickering LK, Baker CJ,Kimberlin DW, Long SS, eds. Red Book: 2009Report of the Committee on Infectious Diseases. 28thed. Elk Grove Village, IL:
American Academy ofPediatrics
; 2009:314-321.
3. Markowitz LE, Dunne EF, Saraiya M, et al;Centers for Disease Control and Prevention (CDC),Advisory Committee on Immunization Practices(ACIP). Quadrivalent human papillomavirus vaccine:recommendations of the Advisory Committeeon Immunization Practices (ACIP).
MMWR RecommRep
. 2007;56(RR-2):1-24.
4. Centers for Disease Control and Prevention(CDC). Preventing pneumococcal disease amonginfants and young children: recommendations of theAdvisory Committee on Immunization Practices(ACIP).
MMWR
. 2000;49(RR-9):1-38.
5. American Academy of Pediatrics. Human immunodeficiencyvirus infections. In: Pickering LK,Baker CJ, Kimberlin DW, Long SS, eds.
Red Book:2009 Report of the Committee on Infectious Diseases
.28th ed. Elk Grove Village, IL:
American Academyof Pediatrics
; 2009:380-400.
6. American Academy of Pediatrics. Immunocompromisedchildren. In: Pickering LK, Baker CJ,Kimberlin DW, Long SS, eds.
Red Book: 2009 Reportof the Committee on Infectious Diseases
. 28th ed.Elk Grove Village, IL:
American Academy ofPediatrics;
2009:72-87.
7. Centers for Disease Control and Prevention(CDC). Guidelines for the prevention and treatmentof opportunistic infections among HIV-exposedand HIV-infected children. Recommendations fromCDC, the National Institutes of Health, the HIVMedicine Association of the Infectious DiseasesSociety of America, the Pediatric Infectious DiseasesSociety, and the American Academy of Pediatrics.MMWR. 2009;58:161-164.
http://aidsinfo.nih.gov/contentfiles/Pediatrics_OI.pdf
. Accessed September2, 2009.
8. Rotateq [package insert]. Whitehouse Station,NJ: Merck and Co, Inc; 2007.
9. Marin M, Güris D, Chaves SS, et al; Centers forDisease Control and Prevention. Prevention ofvaricella: recommendations of the Advisory Committeeon Immunization Practices (ACIP).
MMWR
.2007;56(RR-4):3.
10. Bliss SJ, O'Brien KL, Janoff EN, et al. The evidencefor using conjugate vaccines to protect HIVinfectedchildren against pneumococcal disease.
Lancet
Infect Dis. 2008;8:67-80.
11. Calaminus G, Hense B, Laws HJ, et al. Diphtheria(D) and tetanus (T) antibody values inchildren with acute lymphoblastic leukaemia (ALL)after treatment according to Co-ALL 05/92.
KlinPadiatr
. 2007;219:355-360.
12. Patel SR, Ortin M, Cohen BJ, et al. Revaccinationof children after completion of standard chemotherapyfor acute leukemia.
Clin Infect Dis
. 2007;44:635-642.
13. Emir S, Büyükpamukçu M, Köseoglu V, et al.Varicella vaccination in children with lymphoma andsolid tumors.
Postgrad Med J
. 2006;82:760-762.
14. Masten B, McWilliams B, Lipscomb M, et al.Immune response to hepatitis B vaccine in asthmaticchildren.
Pediatr Pulmonol
. 2003;36:522-528.
15. Clarke P, Powell PJ, Goldblatt D, Robinson MJ.Effect of a fourth Haemophilus influenzae type bimmunisation in preterm infants who received dexamethasonefor chronic lung disease. Arch Dis Child
Fetal Neonatal Ed
. 2003;88:F58-F61.
16. LaRussa P, Steinberg S, Gershon AA. Varicellavaccine for immunocompromised children: resultsof collaborative studies in the United States andCanada.
J Infect Dis
. 1996;174(suppl 3):S320-S323.17. McLaughlin M, Thomas P, Onorato I, et al.Live virus vaccines in human immunodeficiencyvirus-infected children: a retrospective survey.
Pediatrics
. 1988;82:229-233.
18. Lindegren ML, Kobrynski L, Rasmussen SA,et al. Applying public health strategies to primaryimmunodeficiencies: a potential approach to geneticdisorders.
MMWR Recomm Rep
. 2004;53(RR-1):1-29.
19. Boyle JM, Buckley RH. Population prevalenceof diagnosed primary immunodeficiency diseases inthe United States.
J Clin Immunol
. 2007;27:497-502.
20. Centers for Disease Control and Prevention.Reported AIDS Cases in Children http://www.cdc.gov/hiv/topics/surveillance/resources/slides/pediatric/slides/pediatrics.pdf.Accessed September 2, 2009.
21. US Organ Procurement and TransplantationNetwork and the Scientific Registry of TransplantRecipients. 2005 OPTN/SRTR Annual Report. ChapterIV: Pediatric transplantation in the United States,1995-2004.
http://www.ustransplant.org/annual_reports/archives/2005/chapter_iv_AR_cd.htm
.Accessed September 1, 2009.
22. American Cancer Society. Detailed Guide:Cancer in Children. What Is Childhood Cancer?
http://www.cancer.org/docroot/CRI/content/CRI_2_4_1X_Introduction_7.asp?rnav=cri
. AccessedSeptember 1, 2009.
23. Moss W, Lederman H. Immunization of theimmunocompromised host.
Clin Focus PrimaryImmune Defic
. 1998;1:1-8.
24. Centers for Disease Control and Prevention.Recommendations of the Advisory Committee onImmunization Practices (ACIP): use of vaccines andimmune globulins in persons with altered immunocompetence.
MMWR
. 1993;42(RR-4):1-18.
25. Levin MJ. Varicella vaccination of immunocompromisedchildren.
J Infect Dis
. 2008;197(suppl 2):S200-S206.